
BIOPHOTOVOLTAICS
Besides organic and inorganic semiconductors, that have reached practical efficiencies and commercial applications, the utilization of photosynthetic material in artificial device to convert light energy is principally demonstrated1. Currently there is a growing recognition of the applicability of the biological machinery of photosynthesis as the active solar cell material. The primary advantage being that these molecular systems have been already optimized for light harvesting and charge separation by over 3 billion years of evolution. In theory the use of photosynthetic proteins may result in biophotovoltaic devices with open circuit voltages in excess of 1 V and non-limiting electron transfer rates1. The utilization of photosynthetic proteins as solar cell material may ultimately provide the cheapest possible photovoltaics devices since these key building blocks can be harvested from organisms such as algae in large scales. Biophotoelectrode designed for photovoltaics may also be applied for chemical fuel production2 and for novel sensing concepts.
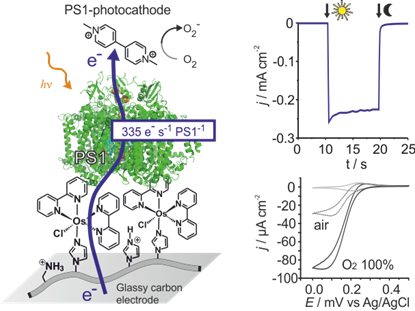
To these ends, we design redox hydrogels for highly efficient electrical contacting and stabilization of the natural photosynthetic protein in artificial devices. While early devices yielded only low efficiency3, the fine tuning of electron mediators with redox potentials adjusted to the one of the protein resulted in several order of magnitude improvement in both photocurrents4 and voltages5. Together with our cooperation partners (W. Schuhmann, M. Nowaczyk and M. Rögner), we have shown that electrons provided by photosynthetic proteins can be recovered at artificial conducting materials, even faster than in natural photosynthesis itself4. The possibility to further improve these performances raise the expectation to eventually achieve practical efficiency from biophotovoltaic devices to provide electrical energy both in high tech devices, such as biocompatible photovoltaics in smart contact lenses6, and in low tech, “painted” large scale solar cells which opens new routes for the cost-per-Watt race in electrical energy production7.
- Plumeré, N. Nature Nanotech. 2012, 7 (10), 616–617.
DOI: 10.1038/nnano.2012.175 - Zhao F., Conzuelo F., Hartmann V., Li H., Nowaczyk M. M., Plumeré N., Rögner M., Schuhmann W., J. Phys. Chem. B, 2015, 119 (43), 13726–13731.
DOI: 10.1021/acs.jpcb.5b03511 - Kothe, T.; Plumeré, N.; Badura, A.; Nowaczyk, M. M.; Guschin, D. A.; Rögner, M.; Schuhmann, W., Angew. Chem. Int. Ed. 2013, 52 (52), 14233–14236.
DOI: 10.1002/anie.201303671 - Kothe T., S. Pöller, F. Zhao, P. Fortgang, M. Rögner, W. Schuhmann, N. Plumeré Chemistry - A European Journal, 2014, 20, 11029–11034.
DOI: 10.1002/chem.201402585 - Hartmann V.,Kothe T., Pöller S., El-Mohsnawy E., Nowaczyk M. M., Plumeré N., Schuhmann W.,Rögner M., Phys. Chem. Chem. Phys., 2014, 16, 11936-11941.
DOI: 10.1039/C4CP00380B - Ravilious K, Clothes that recharge your laptop – the near-endless possibilities of artificial leaves.
Horizon, The research & innovation magazine of the European commission. 16 October 2014 - Plumeré et al., Researchers forecast an Ebola vaccine and low-cost solar cells in 2015.
Horizon, The research & innovation magazine of the European commission, 5 January 2015

