Fakultät für
Chemie
und Biochemie
Biochemie II
Biomolekulare NMR-Spektroskopie
Prof. Dr. Raphael Stoll
Gebäude NC 5/171
Universitätsstraße 150
D-44780 Bochum
Tel.: +49 234 32-25466
Fax: +49 234 32-05466
E-Mail: bionmr@rub.de
Malignant Melanoma
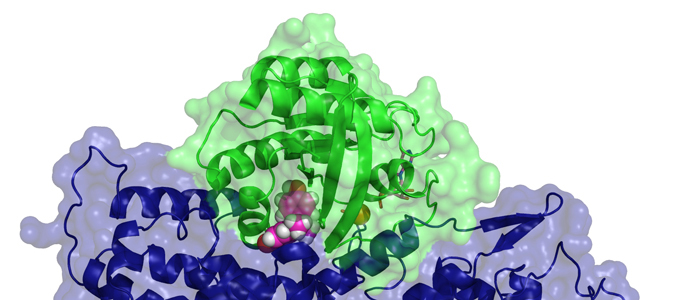
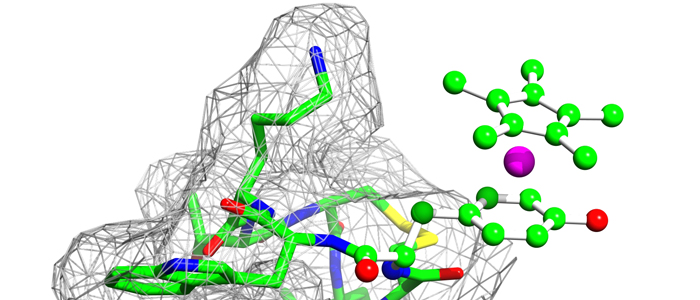
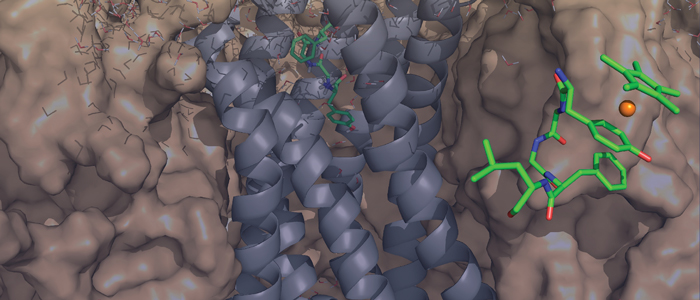
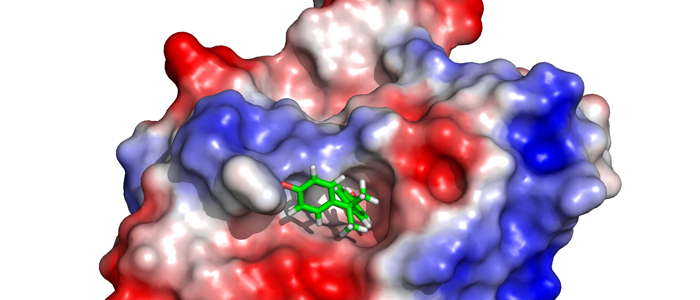
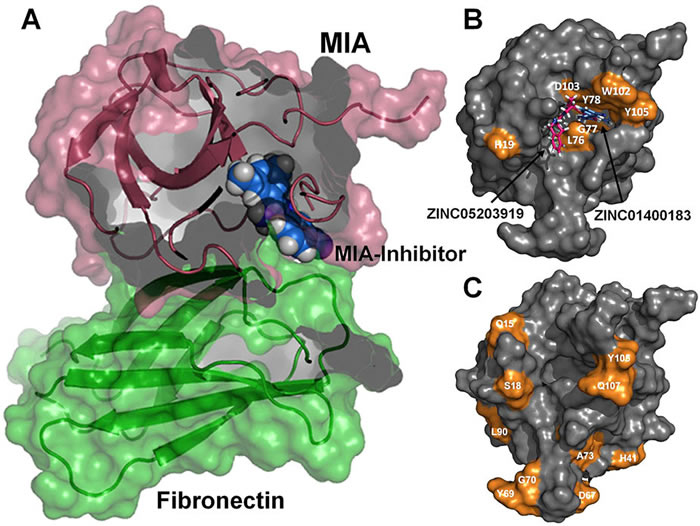
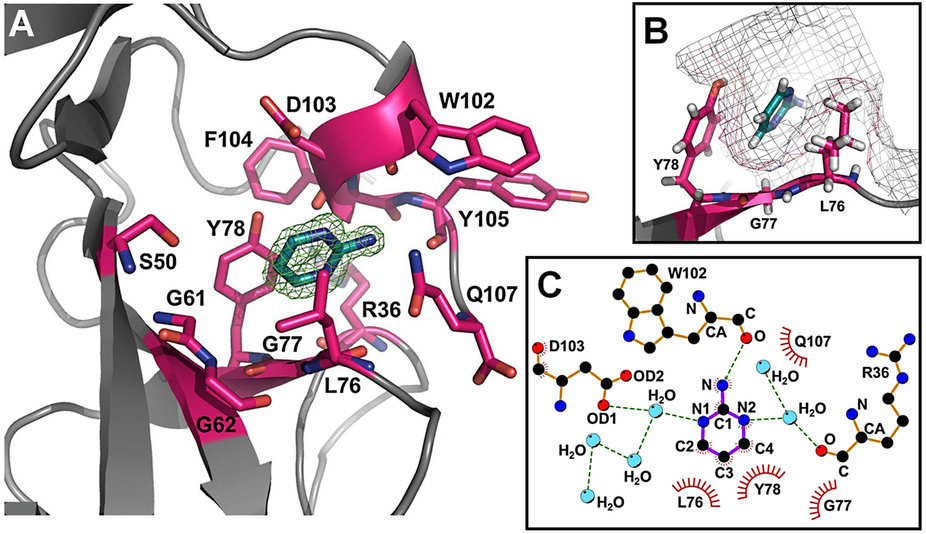 Melanoma inhibitory activity (MIA), an extracellular protein highly expressed by malignant melanoma cells, plays an important functional role in melanoma development, progression, and metastasis. After its secretion, MIA directly interacts with extracellular matrix proteins, such as fibronectin (FN). By this mechanism, MIA actively facilitates focal cell detachment from surrounding structures and strongly promotes tumour cell invasion and migration. Hence, the molecular understanding of MIA’s function provides a promising target for the development of new strategies in malignant melanoma therapy. Here, we describe for the first time the discovery of small molecules that are able to disrupt the MIA-FN complex by selectively binding to a new druggable pocket, which we could identify on MIA by structural analysis and fragment-based screening. Our findings may inspire novel drug discovery efforts aiming at a therapeutically effective treatment of melanoma by targeting MIA.
Melanoma inhibitory activity (MIA), an extracellular protein highly expressed by malignant melanoma cells, plays an important functional role in melanoma development, progression, and metastasis. After its secretion, MIA directly interacts with extracellular matrix proteins, such as fibronectin (FN). By this mechanism, MIA actively facilitates focal cell detachment from surrounding structures and strongly promotes tumour cell invasion and migration. Hence, the molecular understanding of MIA’s function provides a promising target for the development of new strategies in malignant melanoma therapy. Here, we describe for the first time the discovery of small molecules that are able to disrupt the MIA-FN complex by selectively binding to a new druggable pocket, which we could identify on MIA by structural analysis and fragment-based screening. Our findings may inspire novel drug discovery efforts aiming at a therapeutically effective treatment of melanoma by targeting MIA.
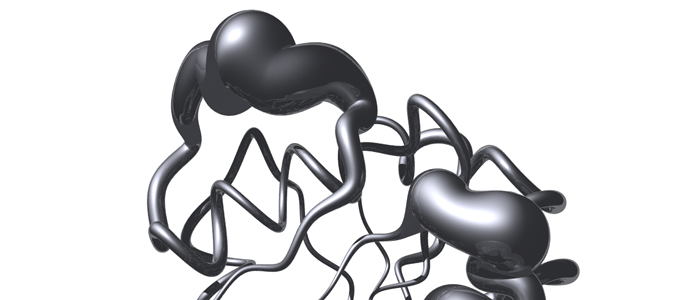
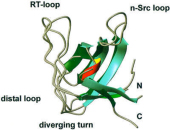 Melanoma inhibitory activity (MIA) protein is a clinically valuable marker in patients with malignant melanoma, as enhanced values diagnose metastatic melanoma stages III and IV. Here we show that the recombinant human MIA adopts an SH3 domain-like fold in solution, with two perpendicular, antiparallel, three- and five-stranded ß-sheets. In contrast to known structures with the SH3 domain fold, MIA is a single-domain protein, and contains an additional antiparallel ß-sheet and two disulfide bonds. MIA is also the first extracellular protein found to have the SH3 domain-like fold. Furthermore, we show that MIA interacts with fibronectin and that the peptide ligands identified for MIA exhibit a matching sequence to type III human fibronectin repeats, especially to FN14, which is close to an integrin alpha4beta1 binding site. The present study, therefore, may explain the role of MIA in metastasis in vivo, and supports a model in which the binding of human MIA to type III repeats of fibronectin competes with integrin binding, thus detaching cells from the extracellular matrix.
Melanoma inhibitory activity (MIA) protein is a clinically valuable marker in patients with malignant melanoma, as enhanced values diagnose metastatic melanoma stages III and IV. Here we show that the recombinant human MIA adopts an SH3 domain-like fold in solution, with two perpendicular, antiparallel, three- and five-stranded ß-sheets. In contrast to known structures with the SH3 domain fold, MIA is a single-domain protein, and contains an additional antiparallel ß-sheet and two disulfide bonds. MIA is also the first extracellular protein found to have the SH3 domain-like fold. Furthermore, we show that MIA interacts with fibronectin and that the peptide ligands identified for MIA exhibit a matching sequence to type III human fibronectin repeats, especially to FN14, which is close to an integrin alpha4beta1 binding site. The present study, therefore, may explain the role of MIA in metastasis in vivo, and supports a model in which the binding of human MIA to type III repeats of fibronectin competes with integrin binding, thus detaching cells from the extracellular matrix.
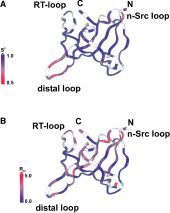 The melanoma inhibitory activity (MIA) protein is a clinically valuable marker in patients with malignant melanoma as enhanced values diagnose metastatic melanoma stages III and IV. Here, we report the backbone dynamics of human MIA studied by 15N NMR relaxation experiments. The folded core of human MIA is found to be rigid, but several loops connecting ß-sheets, such as the RT-loop for example, display increased mobility on picosecond to nanosecond time scales (A). One of the most important dynamic features is the pronounced flexibility of the distal loop, comprising residues Asp 68 to Ala 75, where motions on time scales up to milliseconds occur. Further, significant exchange contributions are observed for residues of the canonical binding site of SH3 domains including the RT-loop, the n-Src loop, for the loop comprising residues 13 to 19, which we refer to as the"disulfide loop", in part for the distal loop, and the carboxyl terminus of human MIA (B). The functional importance of this dynamic behavior is discussed with respect to the biological activity of several point mutations of human MIA. The results of this study suggest that the MIA protein and the recently identified highly homologous fibrocyte-derived protein (FDP)/MIA-like (MIAL) constitute a new family of secreted proteins that adopt an SH3 domain-like fold in solution with expanded ligand interactions.
The melanoma inhibitory activity (MIA) protein is a clinically valuable marker in patients with malignant melanoma as enhanced values diagnose metastatic melanoma stages III and IV. Here, we report the backbone dynamics of human MIA studied by 15N NMR relaxation experiments. The folded core of human MIA is found to be rigid, but several loops connecting ß-sheets, such as the RT-loop for example, display increased mobility on picosecond to nanosecond time scales (A). One of the most important dynamic features is the pronounced flexibility of the distal loop, comprising residues Asp 68 to Ala 75, where motions on time scales up to milliseconds occur. Further, significant exchange contributions are observed for residues of the canonical binding site of SH3 domains including the RT-loop, the n-Src loop, for the loop comprising residues 13 to 19, which we refer to as the"disulfide loop", in part for the distal loop, and the carboxyl terminus of human MIA (B). The functional importance of this dynamic behavior is discussed with respect to the biological activity of several point mutations of human MIA. The results of this study suggest that the MIA protein and the recently identified highly homologous fibrocyte-derived protein (FDP)/MIA-like (MIAL) constitute a new family of secreted proteins that adopt an SH3 domain-like fold in solution with expanded ligand interactions.