Faculty of Chemistry
and Biochemistry
Biochemistry II
Biomolecular NMR Spectroscopy
Prof. Dr. Raphael Stoll
Building NC 5/171
Universitätsstraße 150
D-44780 Bochum
Tel.: +49 234 32-25466
Fax: +49 234 32-05466
E-Mail: bionmr@rub.de
Welcome to Biomolecular NMR at RUB!
Biochemistry seeks to understand life at a molecular level by examining the relationship between the structure and function of biomolecules, such as proteins, nucleic acids, and lipids. To achieve this, we use a variety of techniques, particularly biomolecular NMR spectroscopy, in order to determine the three-dimensional structures and dynamics of biomolecules as well as how they interact with each other and other molecules in solution at near-physiological conditions.
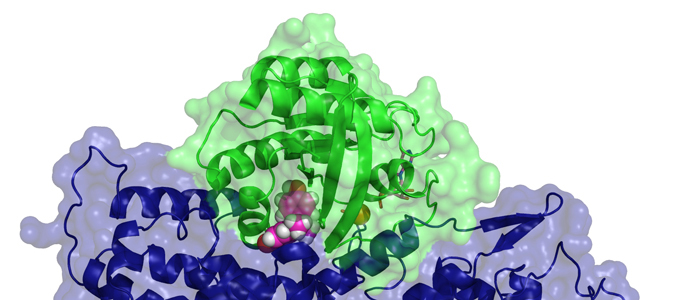
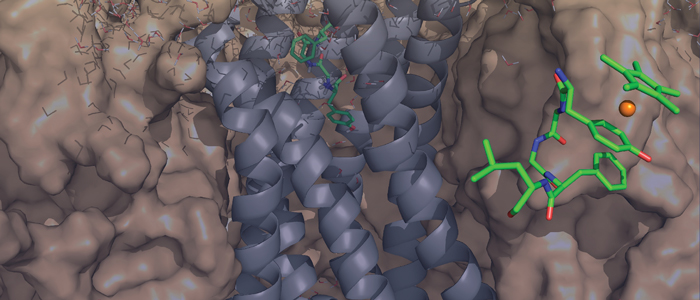
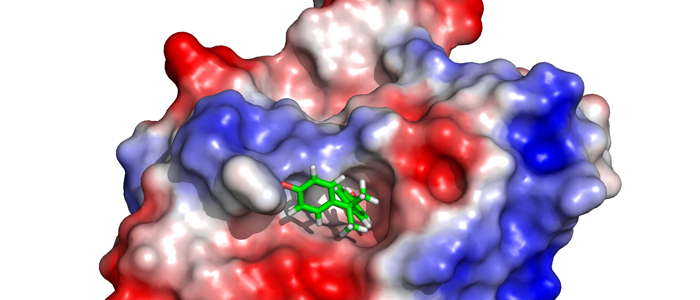
Cancer cells are hallmarked by the ability to divide unrestrictedly, posing an often lethal threat to an organism. Thus, the development of selective small antagonistic ligands tailored to bind to crucial protein targets is paramount to be able to treat cancer effectively. In order to achieve this goal, the molecular mechanism of a potential therapeutic effect needs to be elucidated at atomic resolution.
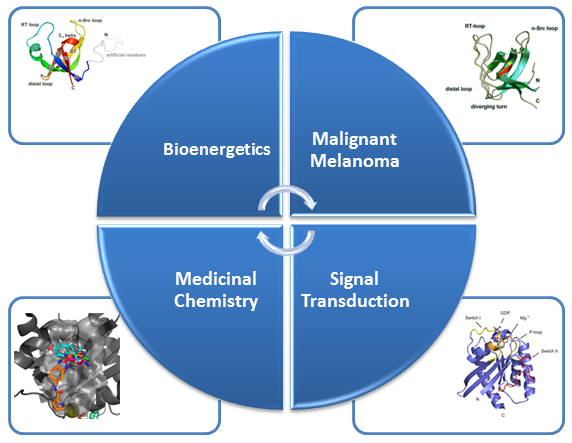
Our research focuses in the main on medically-relevant proteins, specifically those involved in the development of cancerous tumours, to be able to better understand the causes of the condition and propose more effective treatment strategies. Examples include oncogenic proteins, tumour suppressors as well as proteins involved in malignant melanoma, to name but a few. Other current areas of investigation cover the structure, function, dynamics, and ligand interaction of proteins associated with the transduction of physiological signals as well as bioenergetics and infectious diseases, such as tuberculosis.
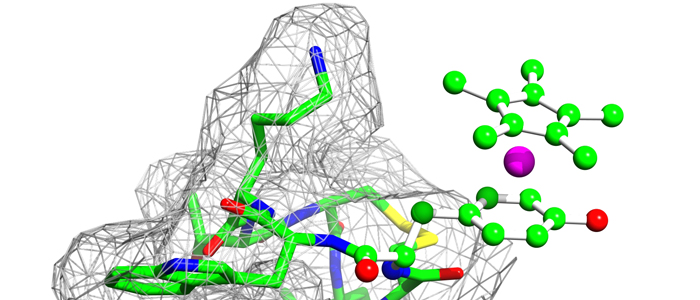
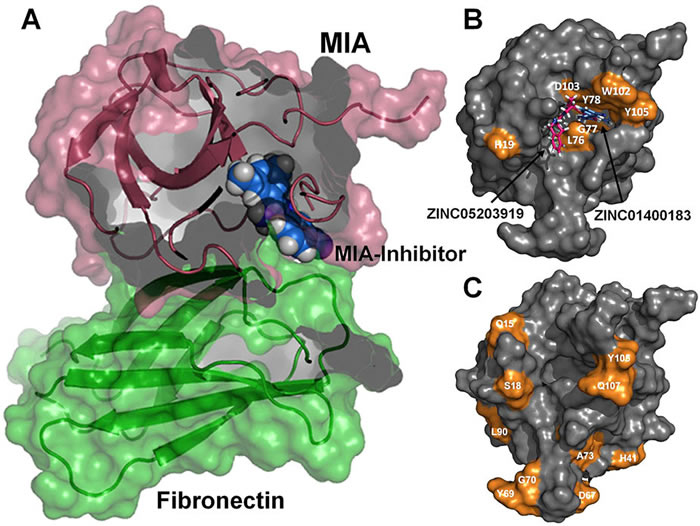
In particular, we would like to understand structure-function-relationships of MDM2, p53, Ras as well as Rheb GTPases, the melanoma inhibitory activity (MIA) protein, NFκB, and HMGA1a. Our additional research efforts in medicinal chemistry include an "SAR by NMR"-like approach in order to develop potential lead structures of small molecular antagonists for these proteins and to combat antimicrobial resistance.