NMR-spectroscopic study of 207Pb in pure and barium diluted lead phosphate
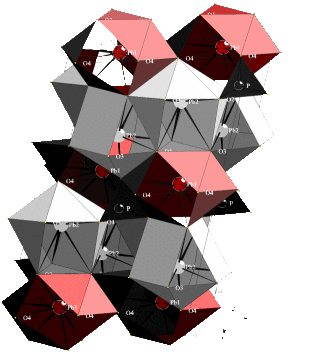
Pure lead phosphate, Pb3(PO4)2, and Ba-diluted samples with chemical composition (Pb1-xBax)3(PO4)2 are well studied improper ferroelastics [1]. The crystals undergo a structural phase transformation from a high-temperature phase with space group R![]() m to a monoclinic low-temperature modification with space group C2/c. The supergroup-subgroup relation allows the formation of three ferroelastic orientation states below the transition point. The dilution of lead by barium leads to strong renormalization effects of the order parameter evolution [2] and to the development of microdomains with typical twin patterns of W and W' walls [3]. The structure of Pb3(PO4)2 was first determined by Keppler [4] and is shown in Figure 1. The structure of the paraphase is characterized by P-atoms tetrahedrally coordinated by oxygen. The tetrahedra are located along the threefold inversion axis, oriented with corners pointing towards each other. Between each set of PO4-tetrahedra are two Pb(2) and one Pb(1) atoms located on the same axis. Cooling the material below the ferroelastic transition point results in a ferroelastic deformation which is primarily accompanied by displacements of all Pb atoms perpendicular to the threefold inversion axis along the binary axis of the ferrophase. The Pb(2) atoms are tenfold coordinated by oxygen neighbours and the Pb(1) atoms are located inside a twelvefold coordination sphere of oxygen atoms [4, 5].
m to a monoclinic low-temperature modification with space group C2/c. The supergroup-subgroup relation allows the formation of three ferroelastic orientation states below the transition point. The dilution of lead by barium leads to strong renormalization effects of the order parameter evolution [2] and to the development of microdomains with typical twin patterns of W and W' walls [3]. The structure of Pb3(PO4)2 was first determined by Keppler [4] and is shown in Figure 1. The structure of the paraphase is characterized by P-atoms tetrahedrally coordinated by oxygen. The tetrahedra are located along the threefold inversion axis, oriented with corners pointing towards each other. Between each set of PO4-tetrahedra are two Pb(2) and one Pb(1) atoms located on the same axis. Cooling the material below the ferroelastic transition point results in a ferroelastic deformation which is primarily accompanied by displacements of all Pb atoms perpendicular to the threefold inversion axis along the binary axis of the ferrophase. The Pb(2) atoms are tenfold coordinated by oxygen neighbours and the Pb(1) atoms are located inside a twelvefold coordination sphere of oxygen atoms [4, 5].
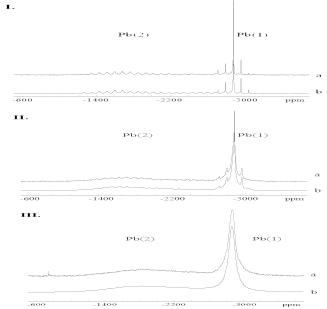
207Pb is the I = 1/2 nucleus with the highest atomic number. The large electron shell gives rise to a resonance frequency which is very sensitive to changes in the local environment of the Pb cation. The known isotropic chemical shift ranges over a wide frequency scale and the chemical-shift anisotropy (CSA) is often large. Hence, spectral widths up to 2000 ppm are common, creating problems of uniform excitation [6]. Thus NMR studies on 207Pb are rare. On the other hand, its sensitivity to its local environment means there is a great deal of structural information in the spectra. The 207Pb MAS spectrum and the corresponding least-squares fit of pure lead phosphate Pb3(PO4)2 can be seen in Figure 2-I.
The two different Pb sites can be distinguished clearly. The superposition of different signals reflects two different crystallographic Pb sites. The first signal near -1700 ppm (Pb(2)) is broad with a width of about 1000 ppm indicating a highly anisotropic shift tensor. The other signal near -2800 ppm (Pb(1)) is about 100 ppm wide. The line widths of the two CSA-MAS pattern sidebands differ. The more anisotropic arrangement of oxygens surrounding Pb(2) leads to a large chemical-shift anisotropy. The more spherical coordination of the Pb(1) causes a small chemical shift anisotropy.
Chemical shift parameters reflect the local symmetry of the oxygen polyhedral environ- ments and refer to the crystal structure. The different isotropic chemical shifts -2881 ppm for Pb(1) and -2017 ppm for Pb(2) correspond to the different Pb coordination numbers of 10 and 12. The integrated intensities of the two resonances give a ratio of 65/35 for the MAS experiment and are in good agreement with the occupancies within experimental error of ![]() . The chemical shift anisotropy of Pb(2) (
. The chemical shift anisotropy of Pb(2) (![]() = -1650 ppm) is about eight times higher than that of Pb(1) (
= -1650 ppm) is about eight times higher than that of Pb(1) (![]() = 201 ppm) and has a negative sign. Compared with other compounds the chemical shift anisotropy for Pb(1) is extremely small. The larger displacement of Pb(2) perpendicular to the threefold inversion axis of is also seen in the higher asymmetry parameter
= 201 ppm) and has a negative sign. Compared with other compounds the chemical shift anisotropy for Pb(1) is extremely small. The larger displacement of Pb(2) perpendicular to the threefold inversion axis of is also seen in the higher asymmetry parameter ![]() . An interest aspect will be structural changes on doping the structure with a larger cation like Ba2+. One of the two Pb sites could be preferentially occupied by Ba. This question has not been clarified up to now in spite of application of different methods like single crystal X-ray diffraction or neutron powder diffraction [3].
. An interest aspect will be structural changes on doping the structure with a larger cation like Ba2+. One of the two Pb sites could be preferentially occupied by Ba. This question has not been clarified up to now in spite of application of different methods like single crystal X-ray diffraction or neutron powder diffraction [3].
Doping of lead phosphate with barium to a mole fraction of x = 0.08 causes an alteration of the chemical shift line shape. Examination of further details of the Pb(1) line shape (Figure 2-II) reveals considerable differences to the resonance generated by Pb(2). The Pb(1) resonance consists of two components: a set of narrow Lorentzian lines, overlapping a second set of wide Lorentzian lines.
The fact that part of the 207Pb MAS signal remains unaltered at this distribution level suggests an inhomogeneous Ba distribution in the structure. This is an agreement with earlier investigations based on optical birefringence. Hensler et al. [3] observed fully extinct areas next to regions with very small optical birefringence in Czochralski-grown single crystals of (Pb1-xBax)3(PO4)2 with x = 0.08 at room-temperature. This optical behaviour displays a spatially heterogenous crystal with the macroscopic ferroelastic transition near room temperature. The coexistence of monoclinic and rhombohedral symmetry at low temperatures (T = 50 - 92 K) was found for single crystals with a barium content of 0.075 ![]() x
x ![]() 0.105. Stronger doping of the structure results in a complete nearing of the Pb(1) CSA line shape. Figure 2-III shows the 207Pb MAS NMR experiment. The Pb(1) signal now consist only of a Lorentzian line shape. The barium content of the structure reaches now a level where it is not possible to distinguish regions with high doping grades and low doping grades from the Pb(1) resonance. The spectrum is consistent with a statistical cation distribution. The larger effect of doping on the Pb(1) site, together with "spin-counting" experiments indicate unambiguously that the Pb(2) sites are preferentially occupied by Ba [7].
0.105. Stronger doping of the structure results in a complete nearing of the Pb(1) CSA line shape. Figure 2-III shows the 207Pb MAS NMR experiment. The Pb(1) signal now consist only of a Lorentzian line shape. The barium content of the structure reaches now a level where it is not possible to distinguish regions with high doping grades and low doping grades from the Pb(1) resonance. The spectrum is consistent with a statistical cation distribution. The larger effect of doping on the Pb(1) site, together with "spin-counting" experiments indicate unambiguously that the Pb(2) sites are preferentially occupied by Ba [7].


